Understanding the Food Safety Modernization Act (FSMA)

The Food Safety Modernization Act (FSMA) is a law mandating the Food & Drug Administration (FDA) to regulate the harvesting, processing, and transporting of food products for human or animal consumption. Passed in 2011, it represents the largest food industry reform in 70 years.
Since then, food and beverage manufacturers and importers have adopted new practices to stay in compliance with new and evolving regulations. To maintain compliance, they must understand the legislation's goals, federal requirements, and how to implement cost-effective automated compliance solutions.
Drawing on decades of experience in food and beverage and other industries that already adapted to enhanced regulations, ifm helps streamline and automate food safety monitoring and documentation while minimizing labor costs and risk.
A paradigm shift for food safety practices
FSMA marks a significant shift from reactive product recalls to proactive, preventive food safety measures. The law:
- Establishes comprehensive, science-based preventive measures to strengthen food safety.
- Expands inspections and compliance to ensure implementation of those protocols.
- Provides the FDA enforcement capabilities to force mandatory recalls.
- Establishes oversight of imported food products.
- Encourages collaboration between the FDA and state or local agencies to build a more integrated food safety system. (Source.)
Since 2013, the FDA has issued 10 rules to implement preventive safety measures from harvest to manufacturing to transportation across the food supply chain. It issued the final rule to establish traceability requirements for high-risk foods in 2024 with enforcement set to begin in January of 2026. Recently, the FDA announced an extension of this compliance deadline to mid-2028 to ensure all entities across the supply chain have adequate time to coordinate sharing of accurate data.
In 1938, the Federal Food, Drug, and Cosmetic Act (FD&C) empowered the FDA to enforce food safety and remove harmful products from the market. It established FDA authority and the current Good Manufacturing Practices (cGMP) requirements for food, additives, drugs, and cosmetics. The DSHEA extended cGMP coverage to dietary supplements in 1994.
How does FSMA enhance previous legislation?
FSMA gave the FDA a mandate to transition from reacting to foodborne illness outbreaks to preventing them. It covers most food production, excluding products regulated under the 1994 Dietary Health and Education Act (DSHEA).
Building on the FD&C Act, the FSMA mandates that the FDA use evidence-based practices to:
- Establish preventive food safety measures
- Inspect food and facilities more often
- Enact mandatory recalls
The FDA established 10 new food safety rules while partnering with state and local agencies to create a nationwide food safety system. It builds on a familiar regulatory framework by using existing definitions from previous legislation to help producers understand compliance requirements.
What is the Hazard Analysis and Critical Control Points (HACCP) framework?
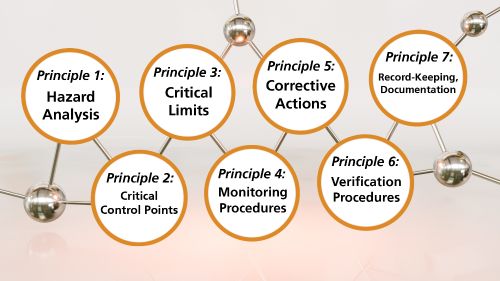
Originally developed by NASA, the Hazard Analysis and Critical Control Points (HACCP) framework is the leading safety protocol for food producers. The seven steps of the HACCP framework are:
- Hazard analysis: identify hazards reasonably likely to cause injury or illness.
- Critical Control Points (CCP): a step where a control can be applied to prevent or eliminate hazards identified in the first step.
- Critical limits: defining the corresponding maximum or minimum values for each CCP to prevent or eliminate the food safety hazard.
- Monitoring procedures: planned oversight of measurements on a continuous or determined frequency ensures CCPs are under control.
- Corrective actions: this establishes procedures for correcting non-compliance at CCPs, how to handle non-compliant products, and record that corrective actions were taken.
- Verification procedures: evaluation, review, and validation that the HACCP plan has identified all hazards and is functioning as intended.
- Record-keeping and Documentation: ensures all established parts of the food safety plan are recorded and responsibilities assigned to team members.
Food producers that use this framework are implementing and establishing best practices for food safety in their production processes.
The FDA recognized its effectiveness in mitigating food safety risks and incorporated these evidence-based practices into the final rules issued under the FSMA.
FSMA Rules and key requirements
The FDA issued 10 final rules to implement the mandate established in the FSMA. These rules and the associated key requirements help food manufacturers, transporters, and importers implement best practices and foster a food safety culture. Almost all rules include some full or partial exemptions. The FDA provides guidance documents for each rule.
Food Traceability Rule
The rule establishes the Food Traceability List (FTL) of high risk foods that must comply with additional traceability requirements. It requires providing the FDA with original, untampered, searchable documentation of traceability data within 24 hours of a recall notice. While focused on high-risk foods, it establishes traceability best practices for the entire industry.
- Read more about the rule and how to comply.
Explore automation solutions:


Preventive Controls for Human Foods Rule
Requires facilities to create and implement a written food safety plan. It uses the HACCP framework, covers preventive control throughout their supply chain, and mandates some elements of previously-optional Current Good Manufacturing Practices (CGMPs). The Rule primarily affects domestic and foreign facilities processing, packing, or holding human foods. It also impacts suppliers and facilities handling by-products for animal food.
- Read more about the rule and how to comply.
Explore automation solutions:
- Smart temperature monitoring
- moneo TT software for traceability of lot codes
- Clean-in-Place real-time phase detection
Preventive Controls for Animal Food Rule
Requires facilities producing animal food products to adopt CGMPs and implement written food safety plans using HACCP framework. The Rule covers all domestic and foreign facilities manufacturing, processing, packing, or holding animal food, including pet food and livestock feed. It currently does not apply to feed mills under different management than the farm and its animals.
- Read more about the rule and how to comply.
Explore automation solutions:


Produce Safety Rule
Establishes safety standards for all operations growing, harvesting, packing, or holding fruits and vegetables for human consumption. It covers agricultural water use, sprouts, soil amendments, worker health and hygiene, animal access to crops, and equipment sanitation.
- Read more about the rule and how to comply.
Explore automation solutions:
- Smart tank level solutions
- RFID solutions for livestock monitoring
- IO-Link for real-time irrigation system monitoring
Pre-Harvest Agricultural Water Rule
Establishes systems-based water assessments for covered produce (excluding sprouts), requiring annual testing and additional assessments when water systems change significantly. It applies to any agricultural operation growing covered produce that uses water for irrigation, cooling, or other pre-harvest activities.
- Read more about the rule and how to comply.
Explore automation solutions:


Sanitary Transportation of Human and Animal Food Rule
Establishes preventive sanitation requirements for human and animal foods during transit by motor vehicle and rail. The Rule applies to shippers, loaders and receivers. It addresses vehicle and equipment cleaning and operations, recordkeeping, and training.
- Read more about the rule and how to comply.
Explore automation solutions:
Mitigation Strategies to Protect Food Against Intentional Adulteration Rule
Requires facilities to implement food defense plans protecting against intentional contamination. It affects large-scale domestic and foreign operations that manufacture, process, pack, or hold food products, and whose products could impact many people if contaminated.
- Read more about the rule and how to comply.
Explore automation solutions:


Foreign Supplier Verification Programs (FSVP) for Importers of Food for Humans and Animals Rule
Requires importers to verify that foreign food suppliers meet U.S. safety standards. It covers all imported food products for humans and animals, including ingredients for further processing. The Rule affects food importers, who must implement verification activities. Very small importers and certain imports from countries with recognized food safety systems may qualify for modified requirements.
- Read more about compliance and requirements.
Laboratory Accreditation for Analyses of Foods (LAAF) Rule
Establishes quality standards for laboratories testing food products. It affects testing facilities seeking FDA recognition to conduct food safety analyses, particularly for imports, environmental testing, and challenge studies.
- Read more about compliance and requirements.


Accredited Third-Party Certification Rule
Establishes guidelines for Recognized Accreditation Bodies to certify auditors for food safety audits and certifying foreign entities. Under this Rule, the FDA can require certification before import when necessary. Foreign companies can expedite product reviews through the Voluntary Qualified Importer Program (VQIP).
- Read more about compliance and requirements.